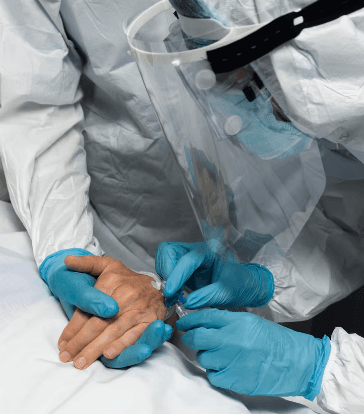
The FDA’s April 9, 2026 early alert on certain Cook Medical sizing catheters is not yet a confirmed recall, but it is already significant news for the U.S. medical and surgical supply market. The agency said Cook identified an increased risk that the marker bands on certain impacted products may crack or break during use. For hospitals and vascular teams, that matters because these catheters are used in angiographic procedures where precise device orientation is important, and any device fragmentation inside the vasculature can quickly turn a routine case into a procedural complication. While the FDA said Cook had reported no serious injuries and no deaths as of April 2, 2026, the agency also warned that worst-case outcomes could include life-threatening harm or death.
According to the FDA, the affected devices include Centimeter Sizing Catheters, Aurous Centimeter Sizing Catheters, and Beacon Tip Centimeter Sizing Catheters. The agency said Cook sent customers a letter on April 2 recommending that the affected products be removed from where they are used or sold. The FDA’s notice instructs customers not to use affected sizing catheters and to return them to Cook Medical. The agency also said the full list of affected product lots is available through the FDA notice.
The issue centers on the catheter marker bands, which help clinicians orient the device within the anatomy during angiographic procedures. The FDA said those marker bands may be at increased risk of cracking or breaking. If that happens during use, the consequences may include longer procedure time, harms associated with device fragmentation or separation, and vessel injury. In a severe scenario, broken material or device separation could result in life-threatening injury. Cardiovascular Business summarized the alert in similarly direct terms, noting that the FDA told providers these catheters should not be used because of the risk of breaking.
This is especially relevant to the surgical supply chain because it affects a device category used in angiographic, aortic, peripheral, and vascular interventions. These are not low-priority stock items. They sit inside procedural workflows where timing, sterility, and product familiarity matter. Even though the FDA has not yet finalized this as a recall, an early alert can still trigger many of the same operational responses: inventory quarantine, case-cart review, supply substitutions, clinician notice distribution, and coordination with vendor representatives. In practice, that means a non-final regulatory action can still create immediate disruption at the hospital level.
The alert also matters because it lands in a device class where physicians rely on consistency and tactile confidence during vascular work. A marker-band defect may sound narrow, but in procedural care, a small component failure can have outsized effects. If a case is delayed while a replacement catheter is located, procedural efficiency suffers. If a catheter fails after insertion, the consequences can expand from simple replacement to added imaging, retrieval efforts, longer fluoroscopy time, and higher risk of vessel trauma. That is why the FDA’s warning is commercially relevant even before a formal recall classification is assigned.
The immediate impact falls on cath labs, vascular surgery programs, interventional radiology teams, materials management departments, and supply chain leaders at hospitals and ambulatory centers. The FDA told customers to examine inventory immediately, quarantine any affected unused product, stop further distribution and use, and return the devices to Cook Medical. The agency also said organizations should share the notice internally and with any outside organizations to which the affected devices were transferred. That means distributors, multi-site systems, and regional supply hubs may need to trace movement across locations rather than simply checking a single storeroom.
There is also a procurement implication. When the FDA uses the early-alert process, it is telling the market that it sees a potentially high-risk device issue and is still reviewing incoming information. That can change purchasing behavior quickly. Health systems may move faster to validate alternative SKUs, ask vendors about available stock depth, and reassess single-source dependence in affected vascular categories. This is an inference based on the FDA’s stated concern and the standard operational response hospitals typically take when high-risk catheter issues emerge.