The U.S. Food and Drug Administration issued an early alert on April 24, 2026, regarding the removal of specific dialysis catheter kits manufactured by Arrow International that contain splittable sheath introducers produced by Merit Medical Systems. The alert highlights a device defect that could lead to sheath failure during vascular access procedures—an issue with direct implications for surgical teams, interventional specialists, and hospital supply chain operations.
Although categorized as an early alert rather than a formal recall classification at the time of publication, the situation has already prompted healthcare providers to reassess inventory, evaluate alternative products, and monitor patient safety risks associated with affected kits.
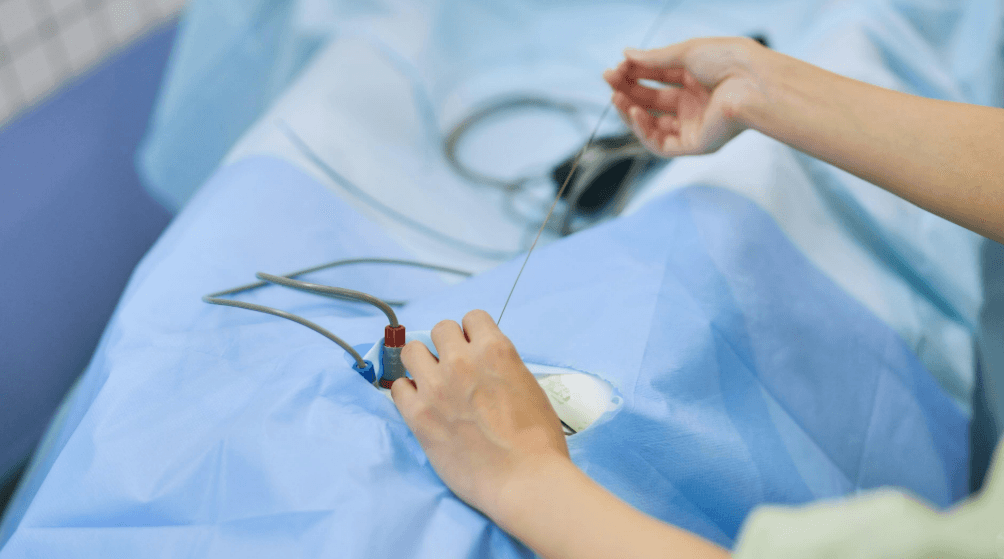
According to the FDA, the affected dialysis catheter kits include a component known as a splittable sheath introducer, which is used to facilitate catheter placement into a patient’s vascular system. The issue stems from a defect that may cause the sheath to malfunction or fail during use, potentially complicating catheter insertion procedures.
The removal was initiated after identifying that the sheath introducer may not perform as intended, creating risks during central venous access. These procedures are commonly performed in dialysis patients, critically ill individuals, and surgical cases requiring reliable vascular access.
The FDA alert advises healthcare providers to immediately review their inventory and quarantine or discontinue use of affected kits. While the agency did not initially assign a recall classification level, the nature of the defect suggests the potential for escalation depending on reported adverse events.
This type of issue is particularly significant because dialysis catheter kits are often packaged as procedure-ready sterile systems, meaning that a defect in one component compromises the usability of the entire kit.
Dialysis catheter kits are widely used not only in nephrology but also in operating rooms, interventional radiology suites, and intensive care units. The splittable sheath introducer is a critical tool that enables safe and controlled vascular access. Any failure in this component can disrupt the procedure, increase complication risk, and extend procedure time.

From a surgical supply chain perspective, the removal introduces several immediate challenges:
• Inventory disruption: Hospitals must identify and remove affected kits, potentially creating short-term shortages
• Product substitution: Clinicians may need to switch to alternative catheter systems, which may differ in handling or compatibility
• Procedure delays: Limited availability of preferred kits can impact scheduling, particularly in high-volume centers
• Cost implications: Emergency sourcing of replacement products may come at higher prices or expedited shipping costs
“According to FDA communications, device component failures during vascular access procedures can lead to complications such as vessel damage, bleeding, or incomplete catheter placement.”
The issue also underscores a broader vulnerability in surgical supply chains: reliance on multi-component kits where a single defective part can render the entire product unusable.
Facilities performing dialysis access procedures, central line placements, and surgical interventions requiring vascular access are directly affected. Supply chain teams must act quickly to identify impacted stock and secure alternatives. ASCs that perform vascular access or related procedures may face similar challenges but often with less inventory buffer, increasing the urgency of replacement sourcing. Interventional radiologists, vascular surgeons, anesthesiologists, and critical care teams rely on consistent device performance. Any variability or failure introduces clinical risk and workflow disruption.
This event reinforces the importance of supplier diversification, inventory tracking, and rapid response protocols. Procurement teams must coordinate with distributors and manufacturers to maintain continuity of care. For Arrow International and Merit Medical Systems, the removal highlights the regulatory and reputational risks associated with device component failures. Distributors must also manage logistics related to product returns and replacements.