MINNEAPOLIS, March 30, 2026 — Medtronic has widened its push in surgical robotics and navigation after winning U.S. Food and Drug Administration clearances that allow its Stealth AXiS platform to be used in cranial and ear, nose and throat (ENT) procedures, extending the system beyond the spine indication it received earlier in 2026. The move gives Medtronic a broader foothold in image-guided and robot-assisted surgery and strengthens its bid to capture more of the company’s targeted $15 billion cranial and spinal technologies market.
The expansion is backed by two separate FDA 510(k) decisions. The agency cleared the Stealth AXiS ENT clinical application on March 16, 2026 under K253395, and then cleared the Stealth AXiS Cranial clinical application on March 26, 2026 under K253379. Those decisions formally broadened the platform’s regulatory scope after its earlier spine clearance in February.
Medtronic says the system combines surgical planning, navigation, and robotics in a single platform designed to support surgeons during complex procedures. In cranial surgery, the company says the platform uses AI-enabled automatic tractography to generate patient-specific brain maps and visualize neural pathways before and during surgery. For ENT procedures, the company says the system is tailored to navigation and visualization of the sinuses and skull base, where anatomy can be tight and high-risk.
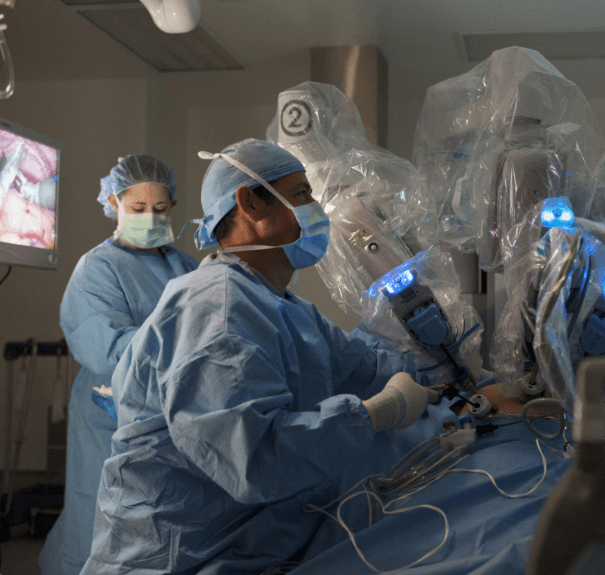
That combination is what makes this clearance commercially important. This is not just an incremental label expansion for a software add-on. It is Medtronic positioning Stealth AXiS as a multi-specialty operating room platform rather than a single-procedure tool. In practice, that changes how hospitals may evaluate the purchase. A platform that can potentially support spine, cranial, and ENT workflows is easier to justify in capital budgeting discussions than a system limited to one service line. It also creates downstream demand for related navigation accessories, service agreements, training, and workflow integration across departments. This is especially relevant for large academic centers and integrated delivery networks trying to maximize use of high-cost OR technology.
Reuters reported that Stealth AXiS combines navigation, imaging, and robotics to guide surgeons during difficult cases, while MedTech Dive emphasized that the platform is part of Medtronic’s effort to expand its share in a large addressable market. Those two points matter because hospitals are increasingly measuring new technology not just by clinical novelty, but by whether it can support higher utilization across multiple specialties. Broader clinical clearance gives Medtronic more leverage in that conversation.
The company is also presenting the platform as part of a larger connected ecosystem. In its March 27 announcement, Medtronic said Stealth AXiS sits inside its AiBLE smart ecosystem and can integrate intraoperative ultrasound through GE HealthCare’s bkActiv system. That matters because hospitals increasingly prefer platforms that can absorb future software upgrades and connect with existing imaging and navigation infrastructure, rather than requiring standalone tools for each specialty. A more modular and connected design could help Medtronic compete more directly in operating rooms that want standardization without sacrificing specialty-specific capability.
Clinical voices featured in Medtronic’s announcement underscored the intended use case. Dr. Michael Ivan of the University of Miami Health System said more information can improve decision-making in complex brain surgery, while Indiana University’s Dr. Vijay Ramakrishnan said intraoperative navigation is important in minimally invasive sinus and skull-base work because it helps surgeons define anatomy in high-risk spaces. Those statements come from Medtronic’s own announcement rather than independent trial results, so they should be read as supportive expert reaction, not comparative proof of superiority. Still, they show how the company is framing the product’s value proposition to surgeons: more precision, more visualization, and smoother workflow.